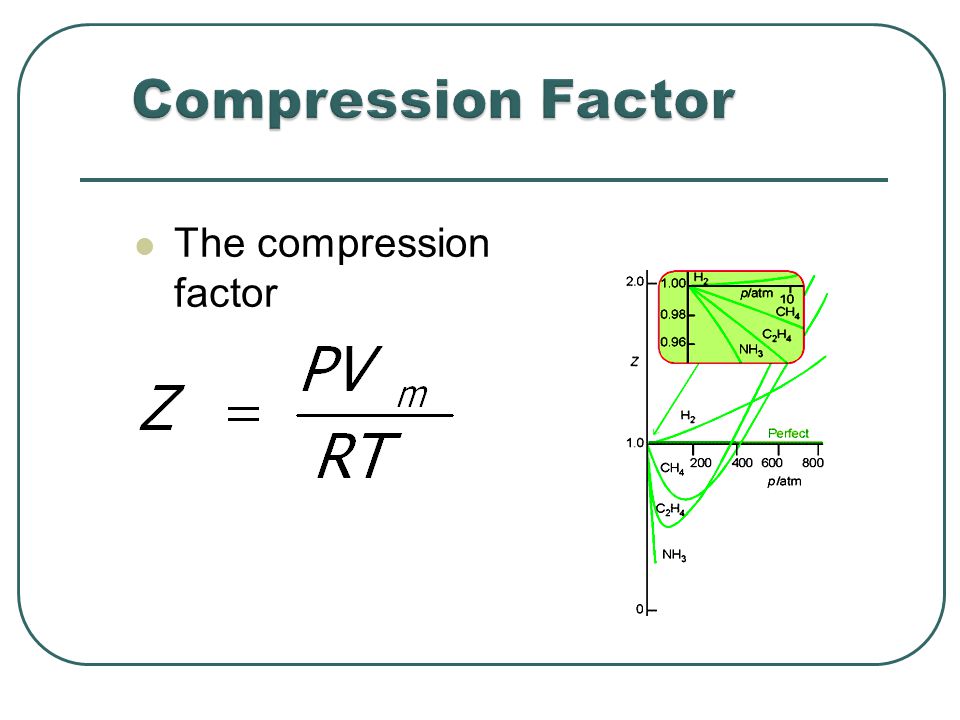
If `Z` is a compressibility factor, van der Waals' equation at low
$ 9.50 · 5 (139) · In stock

If `Z` is a compressibility factor, van der Waals' equation at low pressure can be written as
Answer in Molecular Physics Thermodynamics for Neilmar #278440

Gaseous State Questions for JEE exam - Free Online All questions
20.If Z is a compressibility factor, van der Waals equation at low
Derivation of Van Der Waals Equation

At a high pressure, the compressibility factor (Z) of a real gas is us

What is the compressibility factor (Z) for 0.02 mole of a van der Waals's gas at pressure of 0

Real gasses For an ideal gas, the compressibility factor Z = PV

Compressibility Factor of Gas Overview, Equation & Chart

If Z is a compressibility factor, van der Waals' equation at low press

If Z is a compressibility factor, van der Waals equation at low

If Z is a compressibility factor, van der Waals' equation at low pressure can be written as (2014

16.4: The Law of Corresponding States - Chemistry LibreTexts
The given graph represent the variations of compressibility factor