Researchers discover a way to tease oxygen molecules from carbon
$ 9.00 · 4.6 (426) · In stock
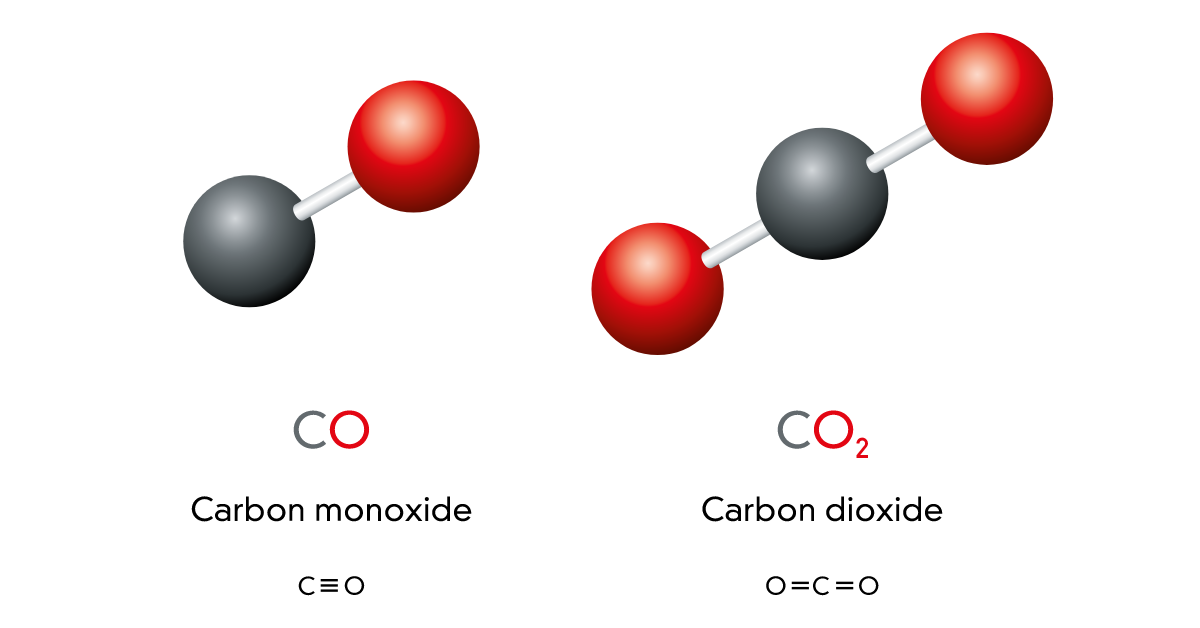
() —A small team of researchers with the University of California has found a way break apart carbon dioxide molecules and get carbon atoms and oxygen molecules instead of carbon monoxide and an oxygen atom. In their paper published in the journal Science, the team describes how they did it, and the implications of their findings. Arthur Suits and David Parker offer a perspective piece in the same journal issue that describes in more depth, minimum energy path (MEP) where reactants don
() —A small team of researchers with the University of California has found a way break apart carbon dioxide molecules and get carbon atoms and oxygen molecules instead of carbon monoxide and an oxygen atom. In their paper published in the journal Science, the team describes how they did it, and the implications of their findings. Arthur Suits and David Parker offer a perspective piece in the same journal issue that describes in more depth, minimum energy path (MEP) where reactants don't always follow the easiest path during chemical reactions and how it pertains to the work done by this group.

Are we alone in the universe?
How to identify oxygen and carbon dioxide if given two jars of
C&EN's Year in Chemistry 2019

How an Asian garden plant led to a fatty acid named after Nebraska

Photosynthesis has unique isotopic signature
How can carbon dioxide be split into carbon and oxygen? - Quora

Nanjing Institute of Geography and Limnology, Chinese Academy of

Bat Guano: A Possible New Source for Paleoclimate Reconstructions
What do you think of the oxygen and carbon dioxide cycle? - Quora
Would carbon sequestration significantly affect the earth's oxygen

EU Research Summer 2023 by Blazon Publishing and Media Ltd - Issuu

NASA Study Finds Life-Sparking Energy Source and Molecule at Enceladus